by Aaron Jonas Stutz

The recently announced human fossil find site of Dinaledi really–and I mean really–defines the phrase of great scientific importance.
Already, the research team led by Lee Berger has uncovered more than 1500 human bone fragments. There appear to be thousands more–perhaps from dozens of individuals–yet to be excavated. The skeletal remains from the Dinaledi site in South Africa will provide us with so much information about an ancient human population that we are sure to confirm certain hypotheses about human evolution … and–of course–overturn others … and this will be done with a shatteringly unprecedented level of confidence about that population’s biological variability.
It’ll just take some time.
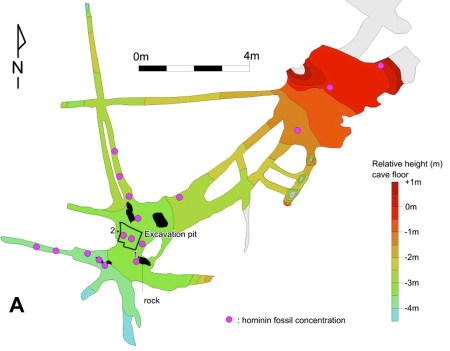
Still, we know it’s going to be hugely scientifically important. In fact, Dinaledi is so huge that the news reports will simply fail to get across how big it is for investigating our shared evolutionary past. (And given how mythically media-sexy the story is–about a dangerous quest deep into the earth to grasp these traces of our past–it’s hard to imagine the media not sufficiently making the case that Dinaledi is “specialer” than other special finds. I’m telling you. Just wait … there’s gonna be more for years to come.)
Despite its sure-thing knowledge-transforming significance, there are two weird challenges with which the Dinaledi finds confront us. And by cloaking the skeletal finds in mystery, these unusual problems are only likely to enhance the site’s scientific staying-power.
Problem 1: Lost in Time
The team excavating and studying the Dinaledi chamber, its sedimentary deposits, and the associated human skeletal remains has set as priority one this goal: figuring out how old the site is. This brings us to the first problem. They have no immediate clues how old the human remains really are (Dirks et al., 2015). In general, extant cave systems in the Cradle of Humankind region of South Africa began trapping fossils–along with sedimentary debris–some time before 3 million years ago, in the middle of the Pliocene Epoch (ca. 5.3-2.6 mya). However, the Dinaledi chamber has likely been continuously accessible from the surface–albeit through long, narrow, winding dark passages. The human remains could be from a much more recent human population.
The authors of the technical paper on the depositional and geological context of the Dinaledi find site conclude their presentation as follows (Dirks et al., 2015):
This leaves the important question of how old the H. naledi remains are. At this point we do not want to speculate on the age of the deposit considering the reworked nature of the sediments resulting in mixed stratigraphic signatures that impede faunal dating of the fossil rodent remains, and the limited amount of clean flowstone suitable for U-Pb dating (Pickering et al., 2011a, 2011b). Further method development is underway to circumvent this problem.
We just gotta hang on and wait.
And even then, expect controversy and scientific uncertainty to prevail for a while, at least until specialists in radiometric dating come up with some clever chemical and physical measurement solutions that will work, not only for Dinaledi, but for similar cave deposits, too. Basically, in caves where seeping water shapes travertine crusts, trace radioactive isotopes of uranium–and their decay products–get incorporated into the calcium carbonate mineral matrix as flowstones, stalagtites and stalagmites form. And thus, they can provide radiometric clocks to determine when those travertines precipitated. But this only works accurately and precisely if there is not too much detritus (basically, dust, dirt, and pebbles) already with varying amounts of uranium, lead, and thorium involved in what’s known as the U-series of radioactive decay. Notably, the problem of “dirty carbonates” forced the large uncertainty in measuring when calcitic crusts formed on another major fossil find–also from a cave context–that of the Manot calvaria (Hershkovitz et al., 2015).

But Dirks et al. (2015) also observe that the clayey sediments have been somewhat mixed, as occasional water and mud flows in the karstic system moved material around. This not only points to the team’s prudently cautious approach in avoiding any comparisons between the rodent bones from Dinaledi and other African sites of known age. It also brings into focus how few rodent bones were found at all.
And this gets us closer to the second problem. The Dinaledi chamber is so remote and stable, and the fossil assemblage is so thoroughly dominated by human remains, that the only plausible way thousands of bone fragments have gotten into the site is by members of the living group carrying partial or whole human cadavers and depositing them deep under ground.
No cave bears. No underground streams. No crevices under leopard perches. Just human bones distributed across the uneven surface of Dinaledi, accumulating over some millennia, sometime over the past 4 million years.
In fact, the study published by Dirks et al. (2015) is a wonderful model of scientifically rigorous process-of-elimination thinking. They convincingly establish that most of the human remains ended up in Dinaledi because living humans in the past put them there … mainly, if not exclusively, as intact cadavers. This seems to be a very exciting discovery in itself: contextual evidence for repeated intergenerational disposal of the dead.
Problem 2: Imagining the Dead
In other words, there’s evidence for socially and semiotically complex, highly evocative ritual. And perhaps very deep in our prehistoric past.
So why is this a problem? It’s not just that we still lack chronological context for the material traces of such ritualized behaviors that–from a cross-cultural and more recent archaeological perspective–would have been so integral to the shaping and reshaping of the social fabric and social experience of the living (see Nilsson Stutz, 2003). It’s that these mortuary ritual acts are documented through the deposits of MANY fragmentary skeletons, which exhibit anatomical features unambiguously unique to the genus Homo, but very definitely small-brained Homo (ca. 500 cubic centimeters).
Our big brains (roughly three times as big) are not necessary conditions for our intensely socially focused, semiotically rich (that is, intensely symbolically attention-focusing, likely recursively evoking) interaction and embodied experience with the world.
And at first glance–and notice the almost unavoidable visually metaphorical expressions we tend to use when we use language to manage each other’s joint attention–this is a bigger scientific problem than that posed by the early 20th Century discovery of human fossil evidence, which falsified the assumption that our that big brains evolved early in our ancestors’ divergence from the African apes (classically summarized by Washburn [1951]). This now-discarded claim was manifest over 100 years ago in the fraudulent wish-fulfillment attempt that was the Piltdown skull. But with new evidence from Dinaledi, we can no longer take for granted another untested claim: that big brains and language are needed to carry out meaningful, emotional, memory-shaping, socially bonding rituals.
So we might now wonder, under the right environmental circumstances, might chimpanzees and gorillas–with brain sizes similar to those of the Dinaledi human remains–begin to teach each other how to care for their dead? Have some past or unobserved extant chimp or gorilla societies already developed such social traditions? And what would this entail for our understanding the nature and diversity of hominid embodied mental experience, with and without language?
Now, the adult modern human brain usually takes up over 1200 cubic centimeters, sometimes as large as 1900 cc’s. The adult cranial fragments from this new report (Berger et al., 2015) are only consistent with brains similar in size to australopithecines and the earliest diagnostic members of the genus Homo from Africa, dating to 3 million to 1.5 million years ago–that is, around 500 cc.
The remains excavated so far from the Dinaledi Chamber in the Rising Star Cave system (located in the Cradle of Humankind group of caves near Pretoria, South Africa) could be from the earliest phase of fossil deposit formation (ca. 3 million years ago).
But as the chamber has likely been continuously accessible from the surface–albeit through long, narrow, winding dark passages–the relatively small-brained human population represented by the Dinaledi remains could be very recent. We may now consider this possibility because short-statured, even smaller-brained humans have been found from archaeological deposits well-dated to ca. 90-12,000 years ago in Liang Bua Cave on Flores Island, Indonesia (Brown et al., 2004; Morwood et al., 2004, 2005).
In short, the newly reported remains from Dinaledi Chamber could be ca. 3 mya … or … when? 100,000 years ago? 50,000 years ago? 12,000 years ago? The new small-brained human remains–dubbed as the new species (!!!) Homo naledi by the team excavating and studying them–have a place, but they don’t have a time. And this inspires Rorschach-test-like associations. How we handle our initial, seemingly inspired thoughts–that is, how we use our initial reactions to focus on logical, theoretically reasonable, necessary and sufficient chains of implications, tracing branching paths of contingent implications, in order to figure out the most relevant hypotheses and rigorous observational approaches to testing them … That’s the test of our scientific mettle.
So perhaps challenge is a better word here than problem, concerning how living members of the group appear to have ritually disposed of the dead resting in Dinaledi chamber.
But it’s a big challenge. Regardless of when the humans represented by the Dinaledi remains lived … and regardless of whether they really belonged to a distinct (and now extinct) branch of the hominin family tree, we also need to focus more deliberately on our tendency to assume that language-based thinking evolved first … only later allowing socially shared imagination and abstraction, in this case, about relationships between the living self and dead loved ones, relatives, allies, or enemies.
Could it have been the other way around? That imagination evolved first in combination with ritualized social interaction? And spoken language only later?
References
Berger, L. R., Hawks, J., Ruiter, D. J. de, Churchill, S. E., Schmid, P., Delezene, L. K., … Zipfel, B. (2015). Homo naledi, a new species of the genus Homo from the Dinaledi Chamber, South Africa. eLife, 4, e09560. http://doi.org/10.7554/eLife.09560
Brown, P., Sutikna, T., Morwood, M. J., Soejono, R. P., Jatmiko, Wayhu Saptomo, E., & Awe Due, R. (2004). A new small-bodied hominin from the Late Pleistocene of Flores, Indonesia. Nature, 431(7012), 1055–1061. http://doi.org/10.1038/nature02999
Dirks, P. H., Berger, L. R., Roberts, E. M., Kramers, J. D., Hawks, J., Randolph-Quinney, P. S., … Tucker, S. (2015). Geological and taphonomic context for the new hominin species Homo naledi from the Dinaledi Chamber, South Africa. eLife, 4, e09561. http://doi.org/10.7554/eLife.09561
Hershkovitz, I., Marder, O., Ayalon, A., Bar-Matthews, M., Yasur, G., Boaretto, E., … Barzilai, O. (2015). Levantine cranium from Manot Cave (Israel) foreshadows the first European modern humans. Nature, 520(7546), 216–219. http://doi.org/10.1038/nature14134
Morwood, M. J., Brown, P., Jatmiko, Sutikna, T., Wahyu Saptomo, E., Westaway, K. E., … Djubiantono, T. (2005). Further evidence for small-bodied hominins from the Late Pleistocene of Flores, Indonesia. Nature, 437(7061), 1012–1017. http://doi.org/10.1038/nature04022
Morwood, M. J., Soejono, R. P., Roberts, R. G., Sutikna, T., Turney, C. S. M., Westaway, K. E., … Fifield, L. K. (2004). Archaeology and age of a new hominin from Flores in eastern Indonesia. Nature, 431(7012), 1087–1091. http://doi.org/10.1038/nature02956
Nilsson Stutz, L. (2003). Embodied Rituals and Ritualized Bodies : Tracing Ritual Practices in Late Mesolithic Burials. Acta Archaeologica Lundensia, 46. Retrieved from http://www.lunduniversity.lu.se/o.o.i.s?id=12683&postid=21368
Washburn, S. L. (1951). The New Physical Anthropology. Transactions of the New York Academy of Sciences, 13(7), 298–304.